How to Synthesize DMF in One Step

N n-dimethylformamide (DMF) as an important chemical raw material and solvent, is widely used in the fields of dyes, medicine, pesticides, and electronics.
This article reviews the DMF synthesis methods, especially the new technology and methods of n n-dimethylformamide synthesis after 2000. It also discusses the advantages and disadvantages of various synthetic DMF catalysts and synthetic routes.
DMF is n n-dimethylformamide, a kind of colorless, high boiling point, hygroscopic, fishy taste of liquid, is a polar non-proton, a high dielectric quantity of organic solvents.
As an important chemical raw material and dissolving ability strong “omnipotent solvent“, DMF is mainly used in polyvinyl chloride (PVC) and polyacrylonitrile synthesis industry such as washing agent, drug intermediates, the pharmaceutical industry, pesticide industry, dye industry, electronic industry, etc.
N, n-dimethylformamide is also as the carrier of dangerous gases, solvent, and adhesives for fixed phase and drug crystallization in other industries.
DMF can also form complexes with many substances such as hydrochloric acid, trioxide and boron trifluoride, and also has very important industrial significance.
DMF has good thermal stability and chemical stability. When there is no acid and alkali, n n-dimethylformamide will not be decomposed even if it is heated to boiling point.
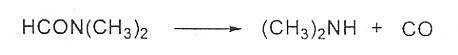
At 350 ℃ or higher temperature, n n-dimethylformamide occurs to decomposition reaction and generates carbon monoxide and dimethylamine. When the aqueous solution containing n n-dimethylformamide hydrolytic boiling happens, it will generate formic acid and dimethylamine.
One-step methods of DMF synthesis
1. carbonylation one-step.
Carbonylation one-step is a method of direct synthesis of DMF by using methanol as a solvent and sodium methanol as a catalyst for the continuous reaction with dimethylamine in high pressure. So far, this method is still the most advanced and economical method in the industry.
Carbonylation one-step method has no water production during the reaction process. And the product can be obtained without water products, and the DMF purity is as high as 98%.
There is no water, the product does not need to separate water. So it saves energy.
Because of dimethylamine with carbon monoxide is in under the condition of direct synthesis of sodium methoxide as a catalyst, raw material price low, wide source, advanced production technology, to save energy and get products of high purity, suitable for large-scale continuous production.
2. Methanol/ammonia/carbon monoxide one-step synthesis
This method greatly simplifies the process of synthesizing DMF, which not only reduces operating costs but also reduces the cost of raw materials.
The reaction was a spontaneous and high response, with a large amount of heat released during the reaction, and its thermodynamic equilibrium constant (0.1mpa, 298K) was 4.81 x 1014.
The strength of acid center and alkali center is the key factor to influence the conversion rate and selectivity.
This article introduces several common DMF synthesis methods. It provides a theoretical basis for further study of DMF synthesis methods and catalysts.
More detail about DMF, please refer to IRO Coating Additive Co., Ltd – www.irocoatingadditive.com
Title



