Project Description
N,N-dimethylformamide, abbreviated as DMF, is a colorless, transparent liquid. It is a polar inert solvent. Except for halogenated hydrocarbons, N,N-dimethylformamide can be mixed with water and most organic solvents.
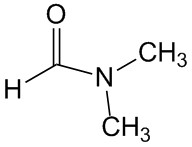
Cas No.:68-12-2
Molecular Formula:C3H7NO
Molecular Weight:73.09
Categories:Aliphatic Nitrogen Compounds
Specifications
| Item | Value |
|---|---|
| Appearance | Transparent, colorless |
| Content | ≥99.9% |
| Color (Pt-Co) | ≤5 Hazen |
| Moisture | ≤0.05% |
| Iron (Fe) | ≤0.050 mg/kg |
| Acidity (HCOOH) | ≤0.001 |
| Alkalinity (DMA) | ≤0.001% |
| Conductivity (25°C, 20% Aqueous), | ≤2.00μѕ/cm |
| pH (25°C,20% Aqueous) | 6.5-8.0 |
Applications
N,N-dimethylformamide is an excellent organic solvent and important chemical raw material. It can be widely used as a solvent for polyurethane, polyacrylonitrile, and polyvinyl chloride.
N,N-dimethylformamide is used in medicine to synthesize sulfadiazine, cortisone, vitamin B6, etc. Pesticides are used to synthesize insecticide squinting. A component used in petroleum chemistry for the extraction of diene.
Synthesis Methods
1.Formic Acid Method
It is generated by formic acid and methanol esterification methyl formate. And then dimethylamine gas instead should be generated dimethylformamide. Then get N,N-dimethylformamide through pressurizing and rectification methanol and methyl formate recovered by distillation.
2. One Step Method
The direct reaction of dimethylamine and carbon monoxide in sodium methanol. Reaction conditions are 1.5~2.5 MPa and 110~150℃. N,N-dimethylformamide can be obtained by rectifying the crude products.
3. High-Pressure Method
Carbon dioxide and methanol under high pressure and 80~100℃ temperature by carbonyl synthesis of methyl formate. Then, the reaction with dimethylamine dimethyl formamide generates N,N-dimethylformamide after distillation.
4. Trichloroacetaldehyde Method
By the reaction of Trichloroacetaldehyde and Dimethylamine, get N,N-dimethylformamide.
Safety Information
Low toxicity, slight irritation to the skin and mucous membranes. But dimethylamine is highly toxic. It can damage the liver seriously. So the production equipment is to be sealed, and the operator is to bring good protective equipment.